|
9/16/2023 0 Comments C2h4 molecular geometry bond angle 
The two sp3 hybridized orbitals overlap and mix with the s orbitals of the hydrogen atoms to produce two N-H sigma bonds. Sp3 hybridization means that the central atoms have hour sp3 hybrid orbitals. This mixing and overlapping occur in alignment with the principles of quantum mechanics which says the atomic orbitals of similar energies can participate in the hybridization process which is inclusive of both filled and half-filled orbitals.įrom the Valence Bond Theory (VBT), it can be analyzed that both the carbon and nitrogen atoms in the methylamine (CH3NH2) are sp3 hybridized as the C-N sigma bond is overlapping between the two sp3 orbitals. Hybridization is a theoretical idea that says the atomic orbitals mix and overlaps in a certain way to form new hybrid orbitals which affect the overall molecular geometry and the chemical bonding properties of the participating atoms changing the complete molecule. This leads to excessive repulsion between the lone pair of electrons and the bonding pair which reduces the bond angle from its ideal percentage. This can be explained with the help of the Valence Shell Electron Pair Repulsion (VSEPR) theory, which says the lone pairs are present in the orbitals that are shorter and rounder than the orbitals having bonding pair of electrons. It might be interesting for you to realize that the ideal bond angle for trigonal pyramidal shape is 109.5° where this deviation from the ideal percentage can be explained by the presence of a lone pair of electrons on the nitrogen atom. Whereas, in the case of nitrogen targeting -NH2 end, its molecular geometry is trigonal pyramidal as the bond angle is around 108.9°. In the case of carbon mainly targeting -CH3, its molecular geometry will be tetrahedral as the forming bond angles are 109.5°. The molecular geometry of a molecule depends upon the Lewis structure and helps with determining the bond length, angles, type, and other geometrical parameters based on the position of each of the participating atoms.Īs there are two central atoms in Methylamine (CH3NH2) the molecular geometry for both will be different as the bond angles are different. Molecular Geometry of Methylamine (CH3NH2) Step 5: Now, draw the structure by analyzing all the aforementioned points: Step 4: Find the type of bond-forming among the atoms of the methylamine (CH3NH2): A single bond is forming among the participating atoms with a lone pair of electrons on the nitrogen atom as methylamine (CH3NH2) is an amine. This makes CH3 and NH2 two separate entities giving rise to two central atoms, that are, carbon and nitrogen. 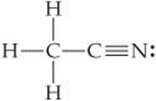
Step 3: Search for the central atom in one methylamine (CH3NH2) molecule: From the molecular formula, it is clear that three hydrogen atoms are bonded to the carbon atom while two are bonded to the nitrogen atom. Step 2: Find for how many more valence electrons are required by one methylamine (CH3NH2): It is 12 for one methylamine (CH3NH2) molecule as 1 valence electron is needed by each hydrogen atom, 4 and 3 are required by the carbon and nitrogen atoms. Step 1: Find the total number of valence electrons methylamine already has: It is 14 for one methylamine (CH3NH2) molecule as 4 are coming from the carbon, 1 from each hydrogen atom, and 5 from the nitrogen atom. Now, let us study the steps involved in drawing the Lewis Structure of methylamine (CH3NH2): Moreover, the electronic configuration of hydrogen and nitrogen are 1s1 and 1s2 2s2 2p1 as their atomic numbers are 1 and 7.Īs s shell can accommodate 2 valence electrons and the p shell can accommodate 6 to stabilize their octet, the valence electron for hydrogen is 1 and that of nitrogen is 5. The electronic configuration of carbon is 1s2 2s2 2p2 as its atomic number is 6.Īs p shell can accommodate a maximum of 6 electrons, there is a vacancy of 4 electrons which makes the valence electrons for carbon 4. To begin with, understanding the Lewis structure of methylamine (CH3NH2) it is first crucial to understand how many valence electrons each participating atom has. When more than one Lewis diagram can be drawn for a molecule, the diagrams are called resonance structures. It is a property conducive to covalent compounds mainly and is used to study formal charge on the participating atoms and chemical bonding taking place between the atoms. To study a chemical compound, the Lewis structure is the first step, to begin with as it helps with determining from which group the molecule belongs in alignment with the periodic table. 
The Lewis structure is a diagrammatic representation of how the movement of valence electrons is occurring to ensure bond formation. Conclusion Lewis Structure of Methylamine (CH3NH2) 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
